TPX-100
TPX-100 is a candidate first-in-class disease modifying osteoarthritis drug (DMOAD).
Current Understanding of OA
It has generally been accepted that OA is a result of aging and excessive wear and tear in the articular cartilage of joints. Impaired function and pain have been attributed to the resulting decrease of articular cartilage and changes in bone underlying the cartilage. Several DMOAD therapeutic candidates have been tested based on this understanding for decades, yet none has demonstrated clinical benefits over and above placebo. For example, recent clinical studies of a chondrocyte growth factor (FGF-18), which showed statistically significant knee cartilage increases, did not demonstrate clinical benefits for patients compared with placebo. The discordance between cartilage increase and clinical benefits has led the FDA to call for input with regard to other structural biomarkers and to replace its 19-year-old guidance for development of OA therapies with a new draft guidance in August 2018.
Recent Developments in the Understanding of OA
Multiple groups of scientists have studied three-dimensional (3D) bone shape change in relation to the pathology of knee OA for over a decade. Using data from a large natural history study with more than 9,000 OA and healthy knees (National Institutes of Health Osteoarthritis Initiative), it has been shown that the bone shape of the knee undergoes unidirectional and irreversible change over time, with a much greater rate of change in osteoarthritic knees than in normal knees. Such accelerated bone shape change starts much earlier than cartilage degeneration and appears to be the most reliable structural marker to date for onset of OA. Further, the extent and rate of the bone shape change predicts progression of OA and need for joint replacement. Bone shape can be measured easily with conventional MRI in three dimensions using an established analytic algorithm, providing an opportunity for objective, quantitative measurement of a promising biomarker for knee OA.
The graphic below shows how the pathological shape changes of human tibiofemoral bones progress.
Courtesy of imorphics, Manchester, U.K.
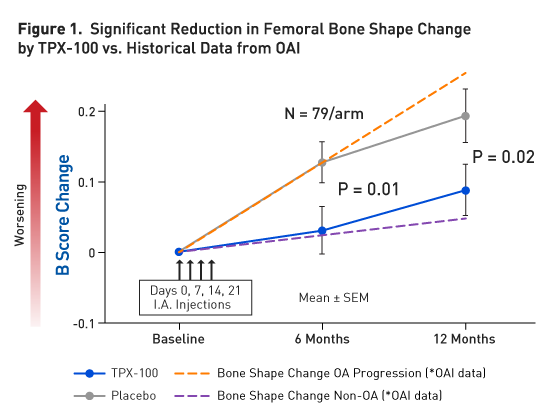
Efficacy of TPX-100 on Knee Bone Shape Stabilization
The bones that comprise a knee joint are the femur, tibia, and patella. The femur and tibia articulate to form the “tibiofemoral” joint, which bears the body weight when standing upright. The femur and patella articulate to form the “patello-femoral” joint which bears up to seven times a person’s body weight when the knee is bent to perform tasks such as going up or down stairs, crouching, squatting, etc. Thus far, femoral bone shape change appears to be the most influential in OA pathology.
In a randomized double-blind placebo-controlled Phase 2 study of
Simultaneous Knee Function Improvement by TPX-100
In parallel with the significant reduction in pathological femoral shape change,
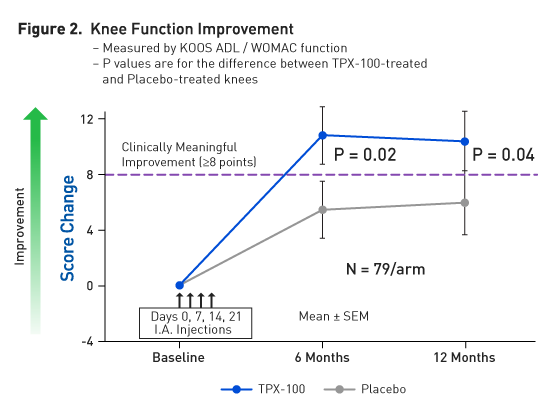
In this clinical study, the test drugs (
Additional Data Supporting OA Disease Modification by TPX-100
Reduction of pathological 3D bone shape changes by
Cartilage Repair Activities of TPX-100
TPX-100 has been known to promote cartilage formation in non-clinical studies prior to the initiation of its Phase 2 studies.
TPX-100 has demonstrated formation of new reparative normal hyaline cartilage in surgical defects in the knee cartilage of Spanish goat model. This model is considered to be especially rigorous in mimicking human cartilage damage and for assessing cartilage repair. The ability of
In the Phase 2 clinical study, only 14% had measurable cartilage change above the limits of detection in patella cartilage, the compartment selected for primary cartilage outcome. Therefore, treatment differences would likely require study times longer than 12 months. However, the Phase 2 clinical study did demonstrate statistically significant correlation between stabilization of tibiofemoral cartilage and critical knee functions at 12 months. Further, stabilization of tibiofemoral cartilage showed statistically significant correlation with reduction of pathological femoral bone shape change. These correlations were not observed in placebo-treated knees. While the striking clinical benefits shown in
Clinical Safety
Over 220 human subjects have received
Compound
TPX-100 is a novel 23-amino-acid peptide derived from matrix extracellular phosphoglycoprotein (MEPE), a 525-amino-acid protein that occurs naturally in humans and is known to be involved in the regulation of hard tissue and phosphate metabolism.
Osteoarthritis
Osteoarthritis, the most common disease of the joints, is one of the most widespread of all chronic, disabling diseases. In the US, osteoarthritis is second only to heart disease as a cause of work disability in men over 50 years of age. Osteoarthritis was the sixth leading cause of years living with disability at a global level, accounting for 3% of the total global years of living with disability. In the US, it is estimated that more than 27 million people suffer from knee osteoarthritis, including more than half of those individuals 65 years of age or older.
Two major health problems with knee osteoarthritis are pain and deteriorating functions in the knees. Typically, patients who start to feel pain and/or knee dysfunction consult their primary care physicians and are advised to try physical therapy and, where appropriate, weight loss to alleviate these problems. They may be advised to take over-the-counter analgesics or are treated with prescription drugs including non-steroidal anti-inflammatory drugs (NSAIDs) and opioids. Once those preliminary treatments become insufficient to control the symptoms, intra-articular therapies may be added. Two major intra-articular therapies are hyaluronic acid products and corticosteroids. Knee joint replacement is often the final option for the most advanced stages of the disease.
There are growing concerns about these conventional approaches to OA therapeutics. Physicians have become cautious about NSAID use because of FDA warnings regarding increased heart attack and stroke risk with even short-term use. The majority of knee OA patients are elderly with substantial risk for these events at baseline, making caution in prescribing NSAIDs prudent. The American Academy of Orthopaedic Surgeons (AAOS) does not recommend hyaluronic acid because of its inadequate efficacy based on clinical trial data (AAOS Treatment of Osteoarthritis of the Knee, Evidence-Based Guideline 2nd Edition). In addition, corticosteroid injections may damage cartilage and bone. The most recent controlled trial of intra-articular corticosteroids in knee OA demonstrated corticosteroid were associated with significantly decreased knee cartilage without significant analgesic efficacy compared with placebo-exposed knees (McAlindon, JAMA, May 2017).
With regard to the treatment of last resort of knee OA, total joint replacement, the cost is roughly $100,000 per knee including post-surgical follow-up and rehabilitation. Artificial knee joints may need to be replaced in 15 to 20 years. Good outcomes to total knee replacement are dependent on rigorous rehabilitation regimes with which many patients are unable to comply. Finally, as with most serious surgeries post-operative complications and morbidities add to the burden of this approach to knee OA.
Clearly, knee osteoarthritis presents a huge unmet need for safe, effective, and affordable therapy to reduce the burden of disease on patients and public resources.
Clinical Merits
TPX-100 is known to act on cells pre-committed to become hard tissue components such as osteoblasts and chondroblasts.
As the FDA stresses in its latest draft guidance, the primary goal for an OA therapy is to “reduce the deterioration of function and worsening of pain” in the patient.





















